
Simplified Ingestion
Seamlessly gather data from upstream systems or manual uploads, purpose-built for regulatory reporting.
Full lineage, version control, and business-owned logic — without rigid pipelines or vendor lock-in.

Collecting data across systems, people, and external sources is overwhelmingly tedious. Validation and preparation for regulatory reporting becomes error-prone and fragile.
Regulatory reports don't exist in isolation. A change in one report can cascade across others — making dependency tracking complex and hard to govern.
Failing to support your submitted figures or respond to ad-hoc requests raises major red flags for regulators and can lead to serious repercussions.
Let's face it — business and regulatory requirements are inherently dynamic. Long implementation cycles often push teams back to manual processes, increasing risk and penalties.

Seamlessly gather data from upstream systems or manual uploads, purpose-built for regulatory reporting.
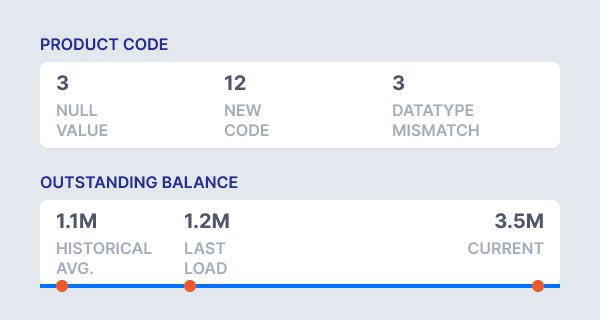
Assess data quality at record and profile levels before validation, ensuring confidence in submitted figures.
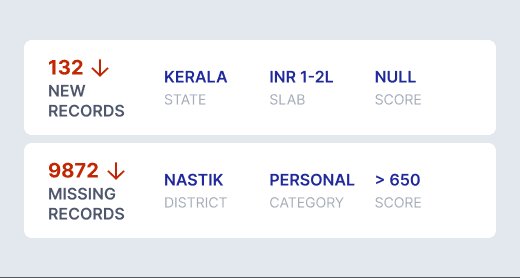
Identify hidden inconsistencies and patterns before submission — reducing regulatory risk and rework.
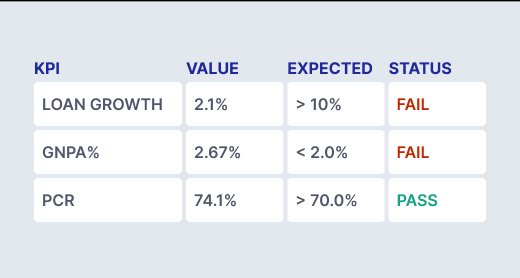
Leverage built-in regulatory validations and KPI frameworks to maintain consistency across reports — even under last-minute changes.
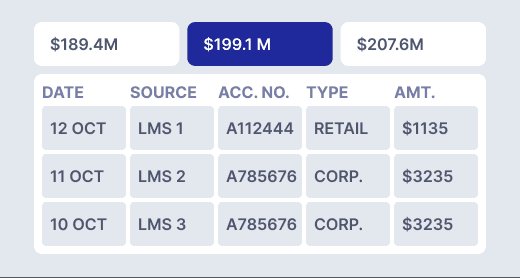
Access detailed breakdowns of reported numbers, enhancing accuracy during submissions and simplifying audits with clear logic and data representations.
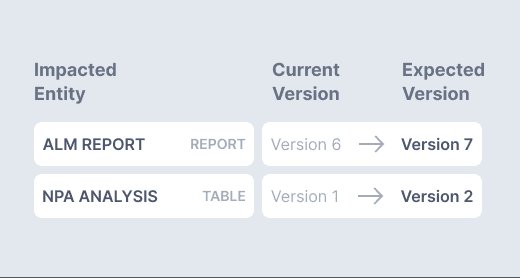
Proactively evaluate potential impact of data changes before submission, holding users accountable for every adjustment.

Complete visibility from raw data to final submission — including enrichments, versions, and commentary.
Create datasets and reports for ad-hoc regulatory requests without reliance on operations or data teams.

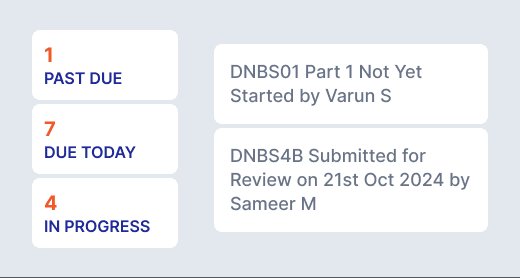
Executive-ready dashboards for monitoring regulatory readiness, submissions, and exceptions across cycles.